
Digital Transformation in Pharma: 2026 Innovation Guide
Quick Summary: Digital transformation in pharma is revolutionizing drug development, manufacturing, and patient care through AI, cloud computing, IoT, and data analytics. Companies like Novartis and Insilico Medicine are achieving 10% productivity gains and reducing drug discovery timelines from years to 18 months. The shift to decentralized trials, smart manufacturing, and real-world data integration is creating a patient-centric industry model that's faster, safer, and more efficient.
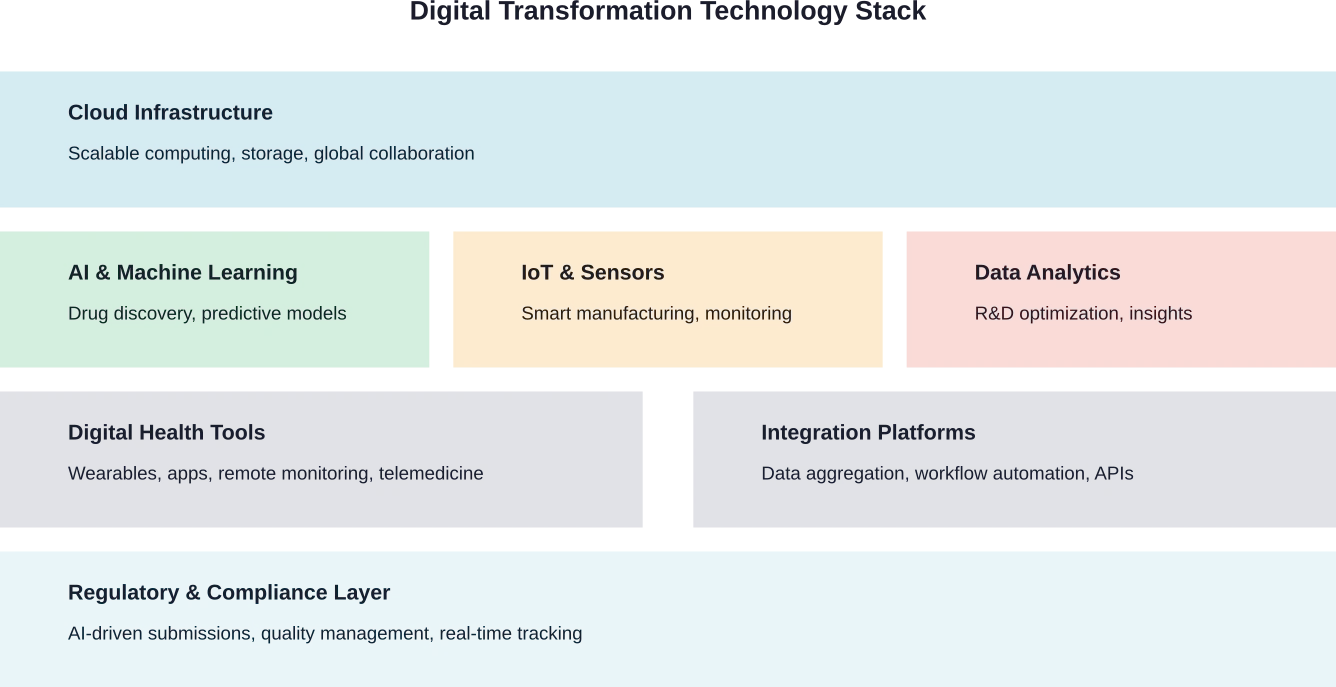
The pharmaceutical industry is experiencing a fundamental shift. What once took years now happens in months. What once required massive physical infrastructure now runs in the cloud. And what once relied on intuition increasingly depends on data.
Digital transformation isn't just about adopting new tools. It's about reimagining how drugs are discovered, tested, manufactured, and delivered to patients. The COVID-19 pandemic turbocharged this evolution—58% of pharma organizations stated that the pandemic has sped up transformation processes within their organisations.
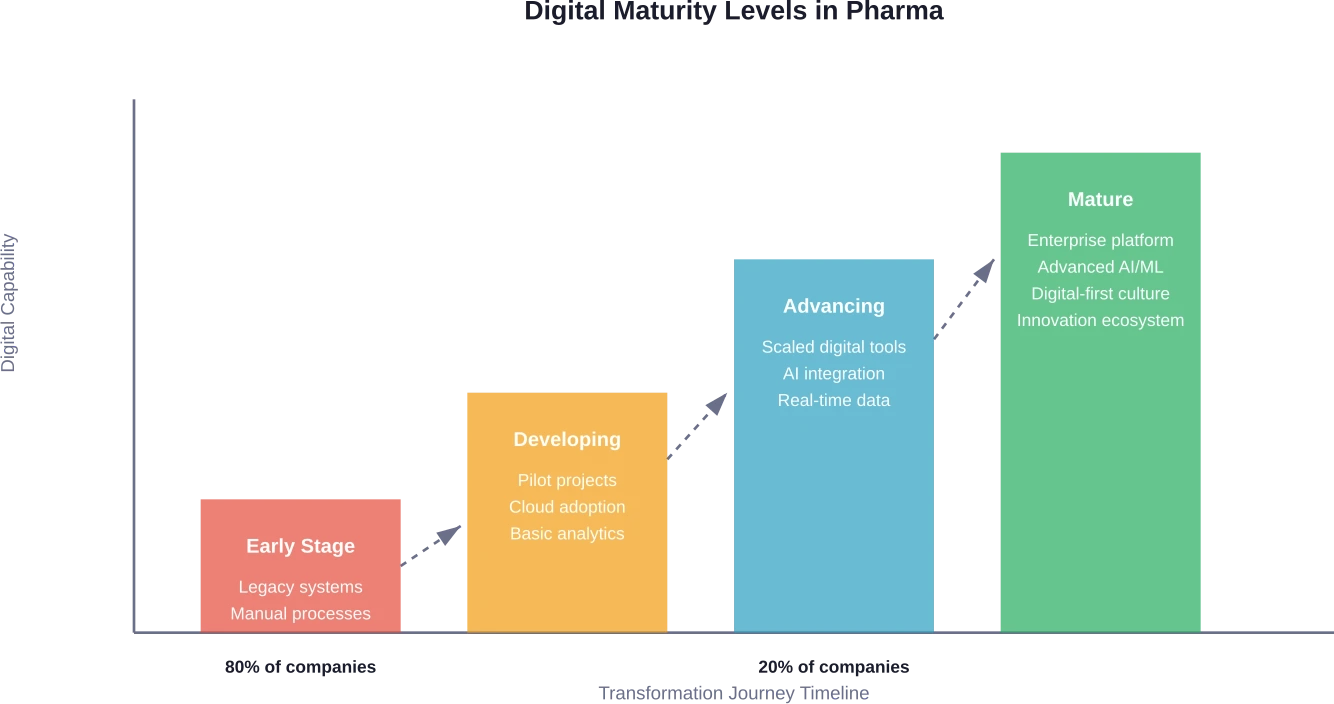
Here's the thing though: adoption remains uneven. According to research, only about 20% of biopharma companies qualify as digitally maturing. The gap between leaders and laggards is widening, and the stakes couldn't be higher.
Why Digital Transformation Matters Now
The FDA recognizes that meeting public health responsibilities requires more effective business processes, increased data capabilities, and coordinated information technology systems. The agency is advancing an enterprise approach to modernization that will optimize use of the vast data foundation underlying regulatory work.
But regulatory pressure is just one catalyst. The business case is equally compelling. Initial projections from digital transformation programs indicate productivity gains in the order of 10% are already achievable, according to research published in Clinical Pharmacology & Therapeutics by Novartis leadership.
Patient expectations have shifted too. Adherence to long-term therapy for chronic illnesses in developed countries averages just 50%—even lower in developing countries. Digital technologies offer solutions through connected devices, mobile apps, and real-world data monitoring that make participation easier and more engaging.
AI and Cloud Computing: Accelerating Drug Discovery
Drug development traditionally follows a brutal timeline. On average, only 0.01% to 0.02% of compounds synthesized in laboratories make it to market. Just 10% of compounds entering clinical development progress to market entry.
Artificial intelligence is changing that calculus. Insilico Medicine's AI system identified a promising fibrosis treatment candidate in under 30 days, with the entire preclinical process from target discovery to IND-enabling studies completed in under 18 months by 2021. The company's AI model designed and validated a preclinical drug candidate in record time by screening molecules and predicting biological activity at unprecedented speed.
GlaxoSmithKline integrated AI into laboratory operations to optimize molecule screening. The system processes millions of potential compounds, identifying candidates worth pursuing while eliminating dead ends early. This approach reduces wasted resources and accelerates the path from hypothesis to clinical candidate.
Cloud infrastructure makes this possible. Processing power that would have required massive capital investment is now available on-demand. Teams collaborate globally on shared datasets. Computational models run continuously, learning and improving with each iteration.

Decentralized Clinical Trials: The Patient-Centric Shift
Clinical trials are moving beyond the traditional site-centric model. Decentralized clinical trials integrate digital technologies and remote processes to enable patient participation from anywhere.
As early as the first year of the pandemic, 76% of sponsors and CROs had integrated decentralized elements into their trials, according to Oracle survey data. The shift addresses a critical pain point: patient adherence and recruitment.
Wearable devices collect continuous data—heart rate, activity levels, sleep patterns—eliminating the need for frequent site visits. Mobile apps deliver medication reminders and capture patient-reported outcomes in real time. Telemedicine consultations replace some in-person appointments.
Research published in Clinical and Translational Science highlights that patient adherence in traditional settings averages just 50%. Digital tools improve that metric by reducing burden and increasing engagement. Participants can continue normal routines while contributing high-quality data.
The FDA and Clinical Trial Transformation Initiative are actively exploring how AI can further improve trial efficiency and accuracy across the drug product life cycle. Regulatory frameworks are evolving to accommodate these innovations while maintaining safety and data integrity standards.
Smart Manufacturing and IoT Integration
Pharmaceutical manufacturing operates under strict quality requirements. A single deviation can compromise an entire batch, costing millions and delaying patient access to critical medications.
Internet of Things sensors monitor every parameter—temperature, humidity, pressure, mixing speeds—in real time. When conditions drift outside acceptable ranges, systems alert operators instantly or adjust automatically. This prevents problems rather than detecting them after the fact.
Digital twins create virtual replicas of manufacturing lines. Engineers test process changes in simulation before implementing them physically, reducing risk and downtime. Predictive maintenance algorithms analyze equipment performance patterns, scheduling maintenance before failures occur.
The result? Fewer batch failures. Less waste. More consistent product quality. And faster response when issues do arise.
|
Technology |
Primary Application |
Key Benefit |
|---|---|---|
|
IoT Sensors |
Real-time environmental monitoring |
Immediate deviation detection |
|
Digital Twins |
Process optimization simulation |
Risk-free testing of changes |
|
AI Analytics |
Predictive maintenance |
Prevent equipment failures |
|
Cloud Platforms |
Multi-site data integration |
Global visibility and control |
|
Robotics |
Automated material handling |
Reduced contamination risk |
Real-World Data and Personalized Medicine
The pharmaceutical industry is sitting on a goldmine of operational data. Electronic health records, insurance claims, pharmacy dispensing records, and now wearable device data create an unprecedented view of how medications perform outside controlled clinical settings.
Real-world data reveals patterns invisible in clinical trials. Which patients respond best to specific treatments? What side effects emerge in broader populations? How do adherence patterns vary across demographics?
Advanced analytics platforms process this information to optimize research and development strategies. Digital biomarkers—measurable indicators captured through sensors and apps—enable personalized treatment approaches. A diabetic patient's continuous glucose monitor data informs dosing adjustments in real time. A cardiac patient's activity and heart rate patterns trigger alerts before symptoms become critical.
This shift from one-size-fits-all to personalized medicine represents a fundamental change in pharmaceutical development philosophy. Treatments are increasingly tailored to patient subgroups or even individuals based on genetic markers, lifestyle factors, and real-world response patterns.

Figure Out Your Pharma System Before You Commit
Digital transformation in pharma is rarely simple - data flows, compliance, internal systems, and external integrations all have to line up. The risk is starting development without a clear structure and ending up with something hard to scale or validate.
OSKI Solutions works at the stage before coding begins. They help define system architecture, data handling, and integrations so you understand what needs to be built and how everything connects. Their work covers custom software, system integration, and data-focused platforms where accuracy and structure matter.
With OSKI, you can:
- map pharma workflows into technical requirements
- plan data flows and system integrations
- get a realistic scope and timeline upfront
If your pharma project feels unclear - sort the structure before you build with OSKI Solutions.
Transform Pharma with Digital Innovation
Modernize pharmaceutical operations with secure and scalable digital solutions. From R&D data platforms and compliance systems to supply chain optimization and analytics, we help pharma companies improve efficiency and innovation.
Data Analytics Platforms: Unlocking R&D Efficiency
Novartis provides a concrete example of analytics platform impact. The company built a suite of digital tools designed to provide insights on patient enrollment, quality metrics, and cost comparisons across studies.
These platforms aggregate data from disparate sources—clinical trial databases, manufacturing systems, supply chain logistics, regulatory submissions. Machine learning algorithms identify inefficiencies, predict timelines, and recommend optimizations.
The transparency changes decision-making. Instead of waiting months for reports, teams access current data dashboards showing real-time progress against targets. When enrollment lags, predictive models suggest interventions before delays compound.
Mobile Apps and Patient Engagement Platforms
Patient engagement extends beyond clinical trials. Pharmaceutical companies are building direct relationships with patients through mobile applications and connected devices.
These apps serve multiple functions. Medication adherence reminders reduce the chronic problem of patients forgetting doses. Educational content helps patients understand their conditions and treatment options. Symptom trackers capture data that informs physician consultations and treatment adjustments.
Some platforms connect patients with similar conditions, creating support communities. Others integrate with electronic health records, ensuring physicians have complete visibility into medication adherence and patient-reported outcomes.
The data flows both directions. Patients benefit from personalized support and better outcomes. Companies gain insights into real-world product performance and patient needs that inform future development priorities.
Implementation Challenges and Solutions
Digital transformation sounds compelling in theory. Implementation is messier. Regulatory constraints, organizational inertia, and unequal digital capabilities create barriers that slow adoption.
Legacy systems pose technical challenges. Data locked in old databases doesn't easily integrate with modern cloud platforms. Validating new systems under regulatory requirements takes time and resources. Training staff on new tools requires significant investment.
Research on the digital divide in pharma highlights that adoption remains fragmented. Companies with mature digital capabilities pull further ahead while others struggle with foundational issues.
But the cost of inaction is rising. Nearly three-quarters of industry professionals agree that the pandemic had the most significant impact on digitalisation efforts. Organizations that delayed transformation found themselves scrambling to enable remote work, virtual trials, and distributed manufacturing oversight.
Successful implementations share common characteristics. Executive sponsorship ensures adequate resources and organizational alignment. Phased rollouts allow teams to learn and adjust before scaling. Partnerships with experienced technology providers accelerate capability building.
Early versions of digital tools are rarely perfect. Initial adopters of these systems encountered bugs, workflow mismatches, and learning curves. But advanced versions would never have been developed without users adopting and providing feedback on initial releases.
Pharma-Startup Partnerships and Innovation Ecosystems
Major pharmaceutical companies increasingly partner with digital health startups rather than building every capability internally. These partnerships provide access to specialized expertise and accelerate time to value.
Startups bring agility and innovation. They experiment with emerging technologies without the constraints of legacy systems or bureaucratic processes. When approaches prove valuable, pharma companies acquire the technology or form strategic partnerships.
This ecosystem approach distributes risk and accelerates learning. Multiple startups explore different approaches to similar problems. Pharma companies observe which solutions gain traction before committing significant resources.
Investment flows both directions. Corporate venture arms fund promising startups. Incubators and accelerators provide mentorship and infrastructure. Joint development agreements align incentives around shared goals.
The Role of Regulatory Bodies
Regulatory agencies aren't passive observers of digital transformation. The FDA actively advances an enterprise approach to business process, data, and technology management that will enable more efficient operations and better use of data.
According to a recent workshop report published in NEJM AI, the FDA and Clinical Trial Transformation Initiative are exploring how AI can be leveraged across drug development while addressing scalability and adoption challenges. The agency recognizes that AI holds immense potential to improve efficiency and accuracy across the drug product life cycle.
Guidance documents are evolving to address digital health technologies, software as a medical device, and AI/ML-based tools. The goal is enabling innovation while maintaining safety and efficacy standards.
International harmonization efforts aim to reduce duplication across markets. When regulatory frameworks align, companies can deploy digital tools globally rather than customizing for each jurisdiction.
|
Challenge Area |
Traditional Approach |
Digital Solution |
|---|---|---|
|
Drug Discovery |
Physical screening of compounds |
AI-powered virtual screening |
|
Clinical Trials |
Site-based patient visits |
Decentralized remote monitoring |
|
Manufacturing Quality |
Periodic batch testing |
Continuous real-time monitoring |
|
Patient Adherence |
Self-reported at appointments |
Connected devices and apps |
|
Regulatory Submissions |
Manual document compilation |
AI-driven data aggregation |
Measuring ROI and Business Impact
Digital transformation requires substantial investment. Justifying that spending demands clear metrics that demonstrate value.
Productivity gains offer the most straightforward measurement. When Novartis reported that initial projections indicated productivity improvements in the order of 10%, that translated directly to cost savings and faster timelines. Fewer resources produce more output.
Time-to-market acceleration has enormous financial implications. Getting a drug to market even three months earlier extends patent protection and captures revenue that would otherwise be lost to generic competition. When AI reduces discovery timelines by years, the cumulative value runs into hundreds of millions per successful drug.
Quality improvements reduce waste. Fewer batch failures mean less material cost and production capacity lost to remediation. Better adherence improves outcomes, reducing the need for intervention therapies or hospitalizations.
But some benefits resist easy quantification. How do you value the competitive advantage of attracting top talent because your digital capabilities enable more interesting work? What's the worth of organizational agility that enables rapid response to market shifts?
Future Trends Shaping the Next Decade
Digital transformation isn't a destination—it's a continuous evolution. Several emerging trends will shape the next phase.
Quantum computing promises to revolutionize molecular modeling and drug simulation. Problems currently requiring weeks of supercomputer time could be solved in hours. Entirely new approaches to understanding protein folding and drug interactions become feasible.
Blockchain technologies may address data integrity and supply chain traceability challenges. Immutable records of manufacturing conditions, custody transfers, and quality tests create audit trails that regulators and patients can trust.
Advanced robotics and automation will extend beyond manufacturing into laboratory operations. Autonomous labs that design and execute experiments based on AI-generated hypotheses could accelerate discovery cycles further.
Synthetic biology tools that allow rapid prototyping of biological molecules will integrate with computational design platforms. The boundary between digital design and physical synthesis blurs.
Frequently Asked Questions
What is digital transformation in the pharmaceutical industry?
Digital transformation in pharma refers to integrating technologies like AI, cloud computing, IoT, and data analytics across drug discovery, development, manufacturing, and patient engagement. It fundamentally changes how pharmaceutical companies operate, making processes faster, more efficient, and data-driven rather than relying on traditional manual approaches.
How much faster is AI-powered drug discovery compared to traditional methods?
AI-powered approaches can reduce drug discovery timelines by approximately 70%. Insilico Medicine identified a fibrosis treatment candidate in under 18 months using AI in 2021, compared to the traditional timeline of 4-6 years for similar discovery stages. However, complete development through approval still requires significant time for clinical trials and regulatory review.
What are decentralized clinical trials?
Decentralized clinical trials use digital technologies and remote processes to enable patient participation without requiring constant site visits. They incorporate wearable devices for continuous data collection, mobile apps for patient-reported outcomes, and telemedicine consultations. By the first year of the pandemic, 76% of sponsors and CROs had integrated decentralized elements into their trials.
What productivity gains can pharma companies expect from digital transformation?
Initial projections from companies like Novartis indicate productivity gains in the order of 10% are already achievable from digital transformation programs. These gains come from faster processes, better resource allocation, reduced waste, and improved decision-making enabled by real-time data analytics. Benefits compound over time as systems mature and organizational capabilities develop.
How do IoT and smart manufacturing improve pharmaceutical production?
IoT sensors provide real-time monitoring of critical manufacturing parameters like temperature, humidity, and pressure. This enables immediate detection of deviations before they compromise product quality, preventing batch failures that cost millions. Digital twins allow virtual testing of process changes, while predictive maintenance prevents equipment failures before they occur.
Why do only 20% of pharma companies qualify as digitally mature?
Most pharmaceutical companies face significant barriers including legacy system integration challenges, regulatory validation requirements, organizational resistance to change, and unequal digital capabilities across the organization. The investment required is substantial, and transformation takes years of sustained effort with executive sponsorship and cultural change management.
How is the FDA supporting digital transformation in pharma?
The FDA is advancing an enterprise approach to business process, data, and technology management to work more efficiently and optimize use of regulatory data. The agency collaborates with industry through initiatives like the Clinical Trial Transformation Initiative workshop on AI in drug development, and develops guidance documents addressing digital health technologies, software as medical devices, and AI/ML-based tools.
Taking the Next Step
Digital transformation in pharma isn't a single project with a defined endpoint. It's an ongoing journey that requires commitment, investment, and cultural change.
The companies leading this transformation aren't necessarily the largest or most established. They're the ones willing to experiment, learn from failures, and iterate rapidly. They invest in talent with digital skills. They partner strategically rather than trying to build every capability internally.
Most importantly, they recognize that the cost of inaction exceeds the cost of transformation. In an industry where timing determines market position and patient lives depend on innovation speed, digital capabilities are becoming table stakes rather than competitive advantages.
The gap between digitally mature companies and the rest will continue widening. The question isn't whether to transform—it's how quickly organizations can build the capabilities required to compete in an increasingly digital industry landscape.
For pharma companies still operating primarily with legacy systems and manual processes, the path forward starts with honest assessment of current capabilities, executive alignment on transformation priorities, and willingness to invest in long-term organizational change. The technology exists. The business case is proven. What remains is commitment to the journey.





